Principal Investigator: Elly Nedivi Massachusetts Institute of Technology Title: “Next generation high-throughput random access imaging, in vivo” BRAIN category: Large-Scale Recording-Modulation – Optimization (RFA NS-14-008)
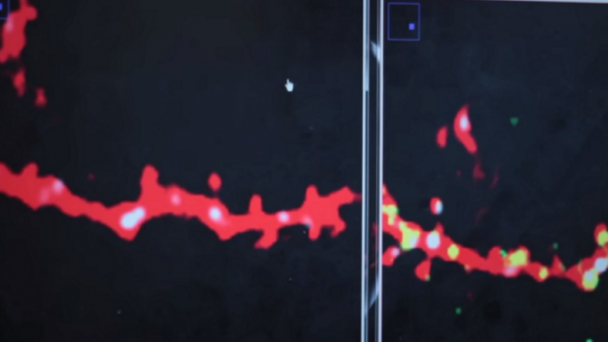
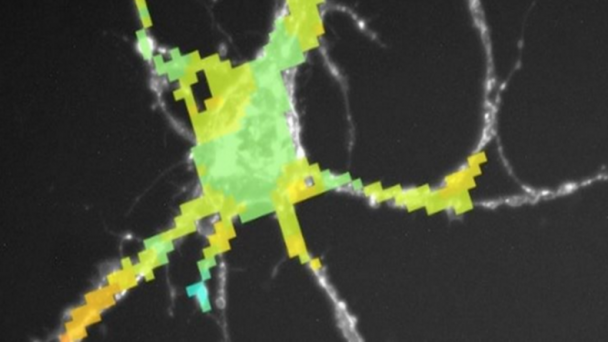
Dr. Nedivi’s team proposes a new imaging technology to simultaneously record activity at each of the thousands of synapses, or communication points, on a single neuron.
https://www.youtube.com/watch?v=lHLSFhp5HawVideo can’t be loaded because JavaScript is disabled: Neurotech 1: Multi-Photon Microscopy (https://www.youtube.com/watch?v=lHLSFhp5Haw)Project Description
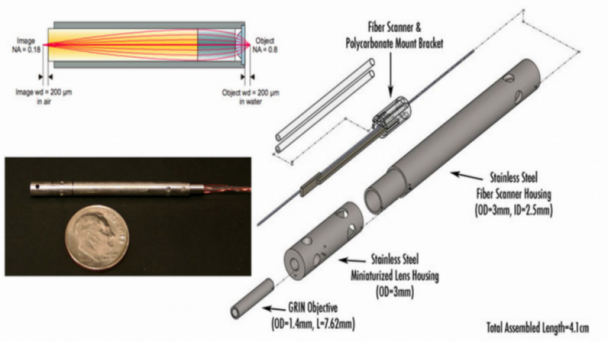
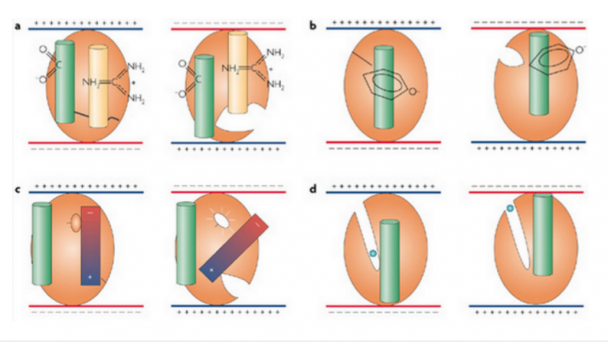
The goal of this proposal is to develop new methods for high speed monitoring of sensory-driven synaptic activity across all inputs to single living neurons in the context of the intact cerebral cortex. Although our focus is on understanding how synaptic inputs are integrated across a single neuron embedded in an intact circuit, the next generation random access imaging technology we propose is more broadly applicable for monitoring multi-cellular activity representing large intra-and inter areal neuronal networks. The approach improves on the speed and sensitivity of current random-access technology by nearly 2 orders of magnitude, enabling high- throughput interrogation of up to 104 independent locations within a fraction of a millisecond and compatible with imaging using next generation voltage sensitive indicators. ...
OnAir Post: Next generation imaging in vivo