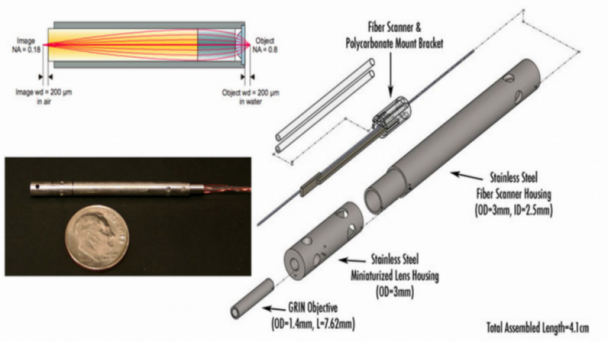
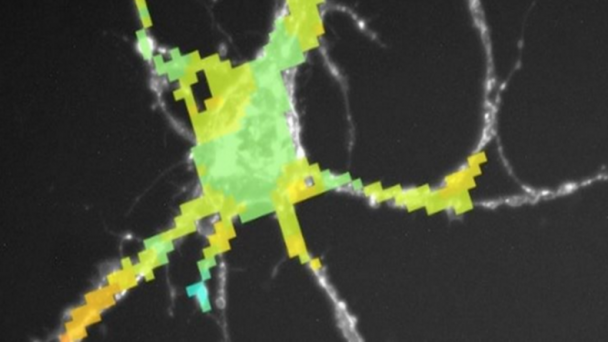
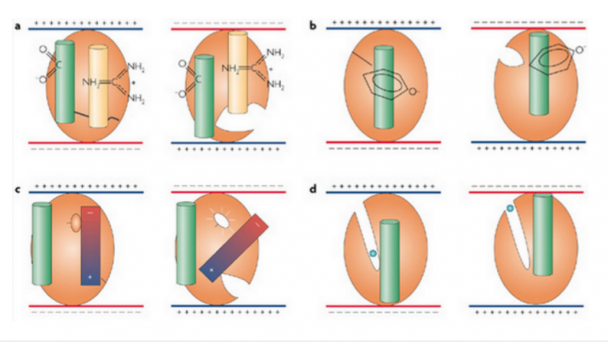
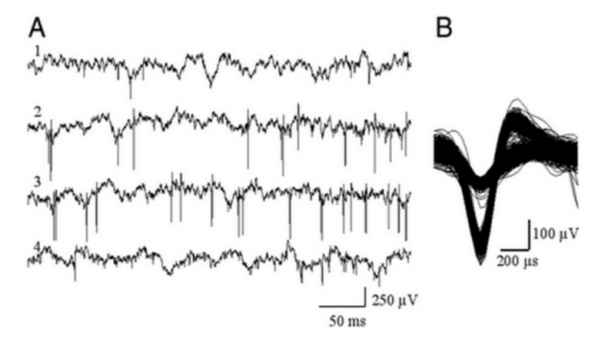
Professor of Physics, Applied Physics, and Bioengineering, CalTech Division of Engineering and Applied Sciences
Director, Roukes Group
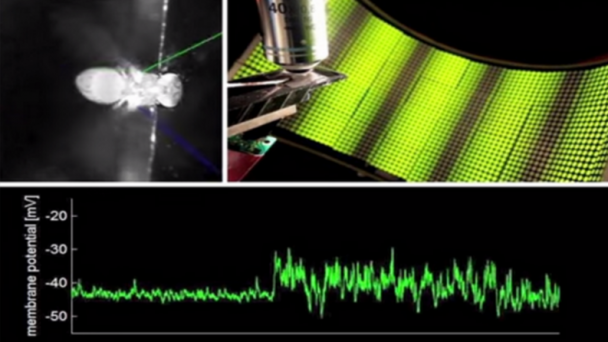
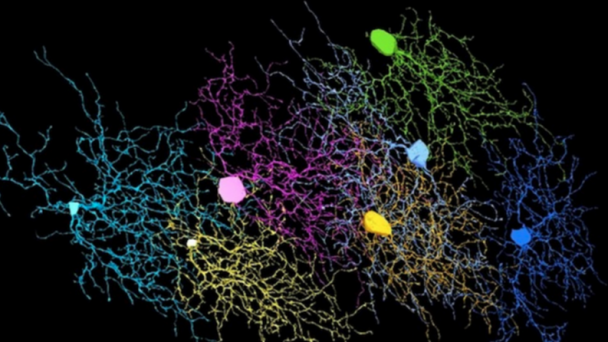
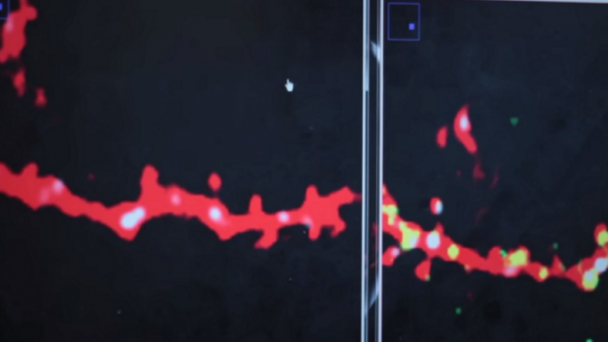
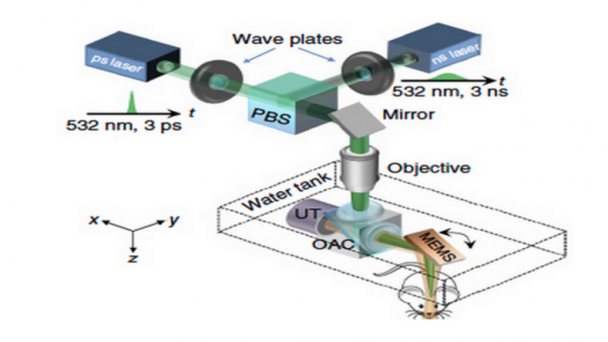
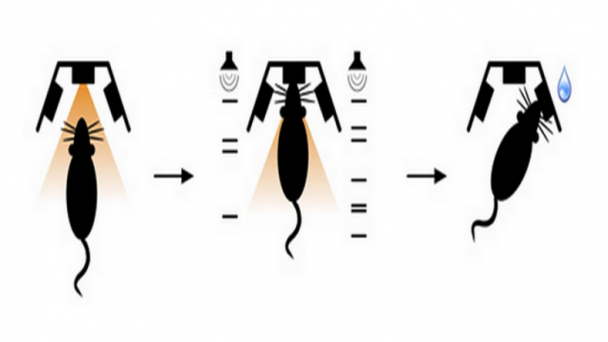
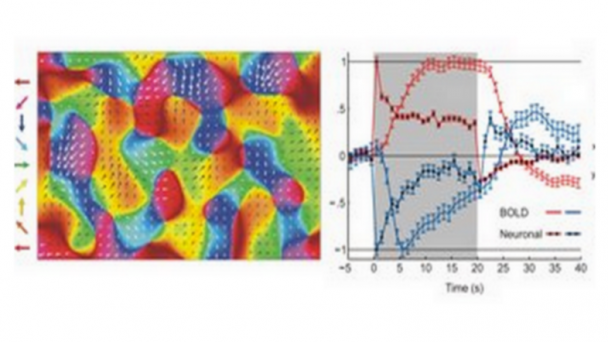
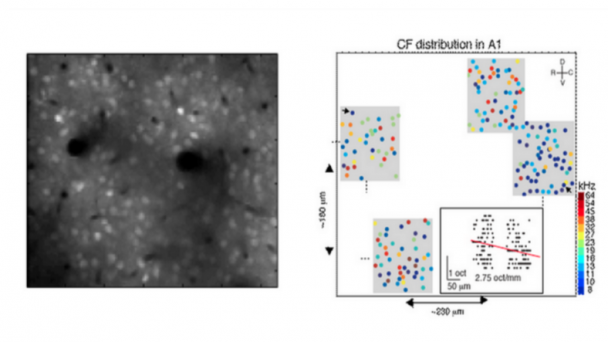
Roukes research activities are currently focused on developing advanced nanodevices, engineering them into complex systems, and using them to enable fundamental problems in neuroscience and proteomics. A continuing thread in theoretical and experimental investigations focuses on fundamental properties of nanomechanical systems.
Web Information
Lab webpage: caltech.edu/people/3185/profile
Division webpage: nano.caltech.edu/people/roukes
Caltech Neuroscience
Brain Initiative Grant
Contact Information
Email: roukescaltech.edu
Phone: 626-395-2916
Address: MC 149-33Pasadena, CA 91125
Biography
B.A., University of California (Santa Cruz), 1978; Ph.D., Cornell University, 1985. Associate Professor, Caltech, 1992-96; Professor of Physics, 1996-2002; Professor of Physics, Applied Physics, and Bioengineering, 2002-11; Abbey Professor, 2011-; Director, Kavli Nanoscience Institute, 2004-06; Co-Director, 2008-2013.
Research
Research Overview
Professor Roukes’s research focuses on nanobiotechnology, nanotechnology, nanoscale physics, nanoscale and molecular mechanics.
List of Research Areas
nanobiotechnology, nanotechnology, nanoscale physics, nanoscale and molecular mechanics
Research Centers
The Kavli Nanoscience Institute, Center for the Physics of Information