Summary
Principal Investigator(s): Steven J. Schiff and Srinivas Tadigadapa Title: Implantable brain microelectromechanical magnetic sensing and stimulation (MEMS-MAGSS) Category: Large-Scale Recording and Modulation Project Number: 1R21EY026438-01 NIH webpage Lab: Center for Neural Engineering University: Penn State Neuroscience
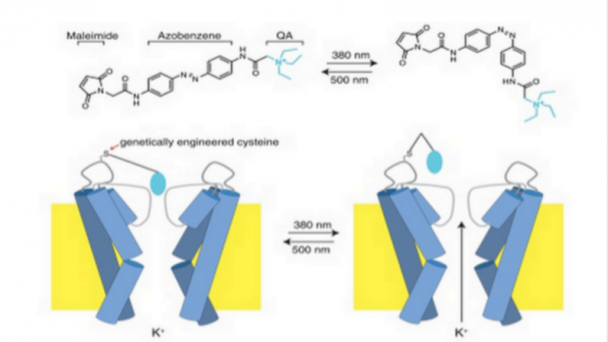
We seek to offer proof-of-concept testing and development of a novel class of MEMS-MAGSS technology. This project would produce a ‘first-of-kind’ technology capable of 1) cellular resolution detection of spiking activity in neurons, 2) cellular level modulation of neuronal firing, 3) adaptve noise cancellation enabling use outside of magnetically shielded environments, 4) room-temperature operation enabling packaging for long-term implantation within with biological tissue for animal or human use, and 5) a clear translational pathway for long-term human implantation across a person’s life-span.
Categories: Penn State Neuroscience, NIH 2015-16 grants, and Research (Neuromodulation)
Project Description
This NIH BRAIN Initiative R21 will initiate development of a completely Implantable Brain Microelectromechanical Magnetic Sensing and Stimulation (MEMS-MAGSS) technology. Significance: We seek to offer proof-of-concept testing and development of a novel class of MEMS-MAGSS technology, to address the NIH BRAIN Initiative: New Concepts and Early-Stage Research for Large-Scale Recording and Modulation in the Nervous System (R21). The current state of the art for large-scale recording of neuronal activity does not have cellular resolution for sensing and ...
OnAir Post: Implantable Brain MEMS-MAGSS